About In Vitro Drug Safety and Biotechnology
Our technology
- Promotes the knowledge of Pharmaco-toxico-vigilance and personalized-precision medicine to avoid drug-adverse events.
- Helps address barriers in medicine and laboratory science to enable the adoption of personalized medicine.
- Benefits patients and the health system.
Innovation
We introduced a personalized approach to the treatment of several chronic diseases:
-
- Alzheimer’s Disease (AD)
- Alcoholic (ALD) and non-alcoholic liver disease (NAFLD)
- Viral hepatitis
- Metabolic diseases (MAFLD)
- Inflammatory bowel disease (IBD)
- Hepato, neuro, entero, cardio and dermato-toxicities
Our Guarantee:
-
- The use of organoid cultures is innovative.
- The laboratory procedure contains a clear rationale.
- The results are compared to standard therapies or/and adequate controls.
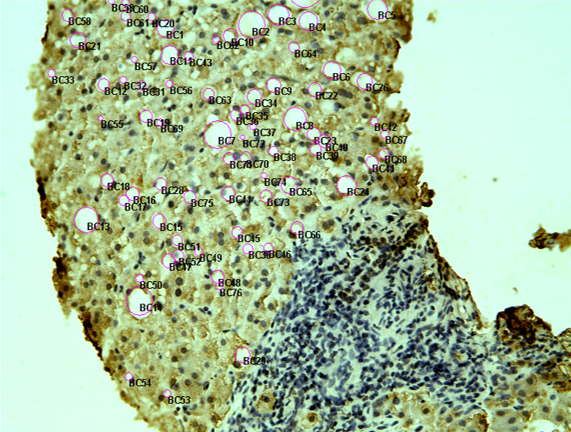
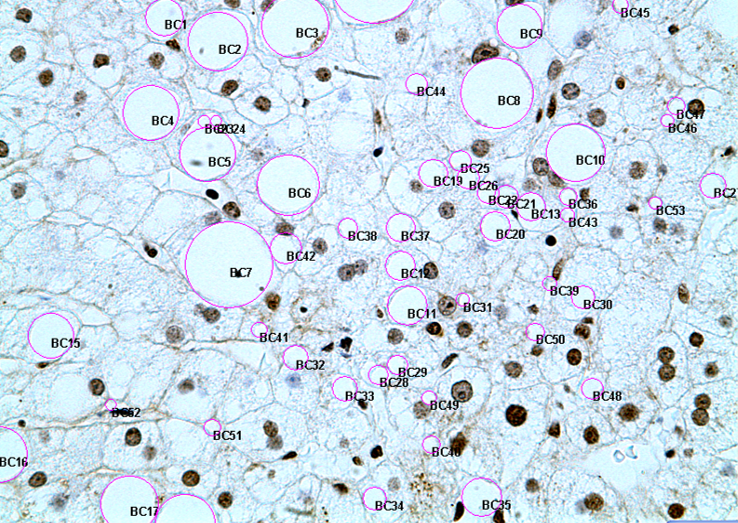
Priorities
- Education on personalized medicine
- Collaboration: In Vitro Drug Safety and Biotechnology has partnered with clinical specialists affiliates with the universities of Toronto, Louisville, Washington, Nebraska, as well as universities in Europe, Israel, Jordan, Australia, Japan and Uruguay
- To introduce non-invasive biomarkers to characterize diseases and to monitor therapies
Dr. Manuela Neuman, Founder and CEO
- Published 25 book chapters, 150 peer-reviewed papers
- Her papers have been cited 19,500 + times.
- Funded for studies in drug-induced liver injury, viral-induced liver disease, inflammatory and autoimmune diseases; alcoholic and non-alcoholic liver disease, neuro and dermatotoxicities as well as inflammatory bowel disease.
- Focuses on nutrition, i.e. vitamins, fatty acids, phospholipids, and methyl donors’ effects on epigenetic mechanisms and liver pathology.
Read more about Manuela’s research and publications
Our Services
In Vitro Drug Safety & Biotechnology is a full-service, clinical reference and research laboratory focused on partnering with hospitals to support their role in community-based medicine.
In Vitro Drug Safety & Biotechnology offers:
- A unified process for ordering and reporting both clinical and anatomic pathology testing services
- A comprehensive menu of both clinical and anatomic pathology laboratory services and cutting-edge molecular diagnostics
- Access to clinical expertise within a network of toxicology specialists and clinical chemists
- Support “special analysis” testing and monitoring clinical trials in: Allergy and Immunology, Clinical and Experimental Pharmacology, Toxicology, Dermatology, Gastroenterology, Genetics, Hepatology, Infectious Disease, Neurology, Oncology, Pathology, Rheumatology
1. Non invasive biomarkers for inflammation in serum and their expression in tissue
- Non invasive biomarkers for inflammation in serum and their expression in tissue
Recent studies strongly suggest that due to the limitations of liver biopsy as well as the improvement of the diagnostic accuracy of biochemical markers, liver biopsy should no longer be considered mandatory to monitor in patients with chronic liver disease.
The markers have specificity for the disease (frontal lobular dementia, Alzheimer’s disease, hypersensitivity syndromes due to different drugs, alcohol-induced hepatitis, non-alcoholic steatohepatitis, drug-induced hepatitis, viral hepatitis B or C alone or in combination with HCV as well as skin diseases).
ELISA and Multiplex analysis
| IL | Interleukins 1, 2, 3, 4, 5, 6, 8, 10, 12, 15, 17, 18, 23, 26, 37 |
| Adiponectin | |
| IFN-alpha, gamma, lambda |
|
| MIP, MPT | Metalloprotease and inhibitor of metalloproteinase |
| RANTES | Regulated on activation normally T-expressed and secreted |
| TLR | Tall receptor 1 and 2 |
| TNF-a | Tumor necrosis factor alpha |
| TNFR1 | Tumor necrosis factor receptor 1 |
| TNFR2 | Tumor necrosis factor receptor 2 |
| VEGF | Vascular endothelial growth factor and its receptors |
Price/analysis 120$ + HST
2. Apoptosis markers
|
AIF |
Apoptosis-inducing factor |
|
AP30 |
Apoptosome |
|
AP-1 |
Activator protein-1 |
|
Apaf-1 |
Apoptotic protease activating factor-1 |
|
Bad |
Bcl-2 family member; pro-apoptotic protein |
|
Bak |
Bcl-2 homologous antagonist/killer |
|
Bax |
B-cell lymphoma X protein |
|
Bcl-2 |
B-cell lymphoma-2 |
|
Bcl-XL |
B-cell lymphoma X protein, long |
|
BID |
BH3-interacting domain death agonist |
|
CAD |
Caspase-activated deoxy-ribonuclease |
|
FADD/FAS-L |
Fas-associated protein with death domain/Fas-ligand |
|
FLICE |
FADD-like interleukin-1 converting enzyme |
|
FLIP |
Fas-associated death-domain like interleukin-1 converting enzyme inhibitory protein |
|
|
Free Light/heavy chain Quantitation in serum |
|
IAP |
Inhibitor of apoptosis |
|
ICAD |
Inhibitor of caspase-activated de-oxyribonuclease |
|
ICE |
Interleukin-1b converting enzyme |
|
M-30/M-65 |
Mitochondrial apoptosis-30/Mitochondrial necrosis 65-factors |
Price/ analysis 190$ + HST
3. Fibrosis markers
The measurements may be used to assess the degree of liver fibrosis and cirrhosis in chronic liver disease and should be recommended as an alternative to liver biopsy.
| HA | Hyaluronic acid |
| TGF-b | Transforming growth factor beta |
| TGF-b R-I | Transforming growth factor beta receptor type I |
| TGF-b R-II | Transforming growth factor beta receptor type II |
Price/analysis 120$ + HST
4. Estrogen and Androgen Receptors
Estrogen receptor alpha and beta
Androgen receptor alpha and beta
Price/analysis 250$ + HST
5. CYP2E1
Cytochrome P450 2E1 – activity and induction needed to produce toxic metabolites of drugs such as acetaminophen or ethanol
Price/analysis 350$ + HST
6. Immuno-histological markers for cytokines and apoptosis
To elucidate the integration of the immune system at the cellular level and programmed cell death in the process of inflammation in disease.
a – Specific Fibriomyalgia test, including the determination and interpretation of 5 different cytokines.
Price/analysis 750$ + HST
b – Neuro-inflammation to distinguish between Fronto-Temporal Dementia and Alzeheimer’s Disease.
Price/analysis 950$ + HST
The work bridges many different levels of analysis including molecular, cellular and cognitive approaches. The cellular and molecular research focuses not only on the underpinnings of normal brain/mind immunologic function an inter-disciplinary research that investigates the role of inflammation and repair as well as the genetic and environmental links to cognitive status.
This analysis will provide a solid basis for further research designed to enhance our understanding of how patients with dementia function, and will hopefully pave the way for future studies that will include the role of of non-invasive biomarkers in monitoring the severity of the disease.
7. Lymphocyte toxicity assay
- Ancillary testing an in vitro lymphocyte toxicity assay is a useful diagnostic tool for severe and life-threatening reactions to drugs such as hypersensitivity syndrome reactions where re-challenging with the culprit drug is not a viable option.
The test has been developed for the fast isolation of human peripheral blood mononuclear cells (PBMCs) from 1-8 mL of freshly drawn anti-coagulated whole blood. Isolated PBMCs are suitable for various applications, such as co-cultures like functional immune cell assays, cytokine expression studies, or immune monitoring of several viral and bacterial infections as well as immune diseases.
In vitro testing in the setting of drug-induced hypersensitivity syndromes as well as defining the pathophysiology of these reactions and aid in the diagnosis and management of other severe drug-induced syndromes. The intention is to develop and perform better diagnostic methods to discriminate between immune and non-immune reactions.
- Assay was validated for
- Sulphonamide antibiotics
- Antiepileptics
- Antipsychotics
- Protease inhibitors
- Antiviral medication
- Non-steroidal Anti-inflammatory
- Steroids
- Anti-gout therapies
- Anti-psoriatic medication
- Biological therapies
- Check-point-inhibitors
- Vaccines
- Immuno-based reactions
- Key markers for defence of the tissue against oxidants: glutathione (oxidized and reduce)
- Detoxification enzymes
- Drug-drug interactions
- Alcohol + drug of use
- Heroin and drugs of use
- Drugs of abuse and anti-psychotics
- Cannabis
The price should be obtained separately for each case. It depends on the number of medications and combination of medications required.
Price/analysis 2500$ + HST
Contact and Shipping
Manuela Neuman Ph.D., FACB
Director In Vitro Drug Safety & BioTechnology
4674 Bathurst Street
Toronto, ONTARIO, CANADA, M2R 1W6
Tel – Office: 416-398-4880
m_neuman@rogers.com
